The modern statement of this relationship, the periodic law, is as follows: the properties of the elements are periodic functions of their atomic numbers. (credit a: modification of work by Serge Lachinov credit b: modification of work by “Den fjättrade ankan”/Wikimedia Commons)īy the twentieth century, it became apparent that the periodic relationship involved atomic numbers rather than atomic masses. Although Mendeleev and Meyer had a long dispute over priority, Mendeleev’s contributions to the development of the periodic table are now more widely recognized ( Figure 2.25).įigure 2.25 (a) Dimitri Mendeleev is widely credited with creating (b) the first periodic table of the elements. The discoveries of gallium (1875) and germanium (1886) provided great support for Mendeleev’s work. But Mendeleev went one step further than Meyer: He used his table to predict the existence of elements that would have the properties similar to aluminum and silicon, but were yet unknown. Both published tables with the elements arranged according to increasing atomic mass. Fluorine (F), chlorine (Cl), bromine (Br), and iodine (I) also exhibit similar properties to each other, but these properties are drastically different from those of any of the elements above.ĭimitri Mendeleev in Russia (1869) and Lothar Meyer in Germany (1870) independently recognized that there was a periodic relationship among the properties of the elements known at that time.  
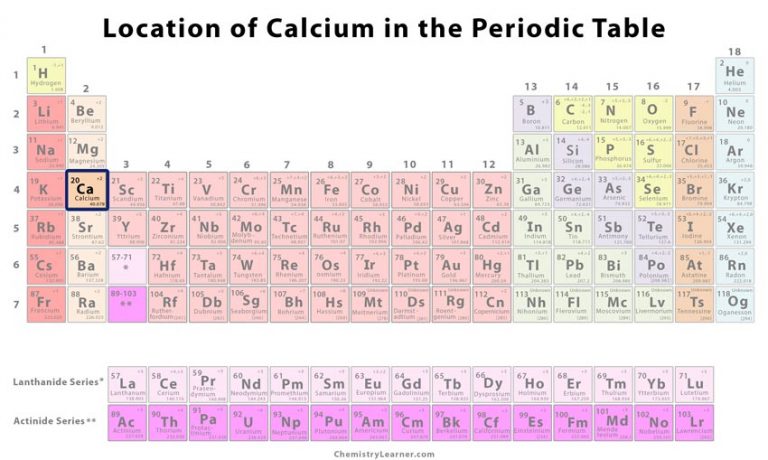
For example: Li, Na, and K are much more reactive than are Ca, Sr, and Ba Li, Na, and K form compounds with oxygen in a ratio of two of their atoms to one oxygen atom, whereas Ca, Sr, and Ba form compounds with one of their atoms to one oxygen atom. However, the specific properties of these two groupings are notably different from each other. A second grouping includes calcium (Ca), strontium (Sr), and barium (Ba), which also are shiny, good conductors of heat and electricity, and have chemical properties in common. One such grouping includes lithium (Li), sodium (Na), and potassium (K): These elements all are shiny, conduct heat and electricity well, and have similar chemical properties. Identify metals, nonmetals, and metalloids by their properties and/or location on the periodic tableĪs early chemists worked to purify ores and discovered more elements, they realized that various elements could be grouped together by their similar chemical behaviors.Predict the general properties of elements based on their location within the periodic table.State the periodic law and explain the organization of elements in the periodic table.One of the most famous compounds is limestone (CaCO 3).By the end of this section, you will be able to: In nature, calcium is always found in compounds with other elements. You won't find calcium sitting around as a pure element. It shouldn't surprise you that calcium has a valence of 2. Those elements in the second column have two electrons ready to make compounds. They all have an outer shell with two electrons and are very reactive. Yes, calcium is defined as a metal because of both its physical and chemical traits. Those elements make up the alkali earth metal family. You will find calcium in the second column of the period table with other elements including beryllium and magnesium. So remember when you look at our breakdown that the electrons aren't always in a nice neat order as shown here. They are found in clouds that can have different shapes that include spheres and dumbbell-like shapes. As you learn more about atomic structure, you will learn that the electrons don't stay in defined areas around the nucleus. The electrons like to be in separate shells/orbitals. In an atom, the electrons spin around the center, also called the nucleus. Each of those colored balls is an electron. If you think this is a little over your head, go back and look at the elements 1-18 that have organizations that are a little more simple. Let's take a look at the arrangements of electrons in the basic elements (left and right sides of the table) of period four and the more complex arrangements of the transition elements (in the middle of the row). You may have an easy way to know the number of electrons in a neutral atom, but the placement of those electrons gets a little more complex. Now we're working with the fourth period/row in the table of elements. 
It tells you the mass of one atom, how many pieces are inside, and where it should be placed on the periodic table. That box on the left has all of the information you need to know about one element. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
